Western Blotting (also called protein immunoblotting) is a laboratory technique used to detect a specific protein within a complex biological sample. While SDS-PAGE separates proteins by size, it can’t tell you which protein is which—many different proteins can be the same weight. Western Blotting solves this by using antibodies that act like heat-seeking missiles, binding only to the one specific protein a scientist wants to study.
How Western Blotting Works
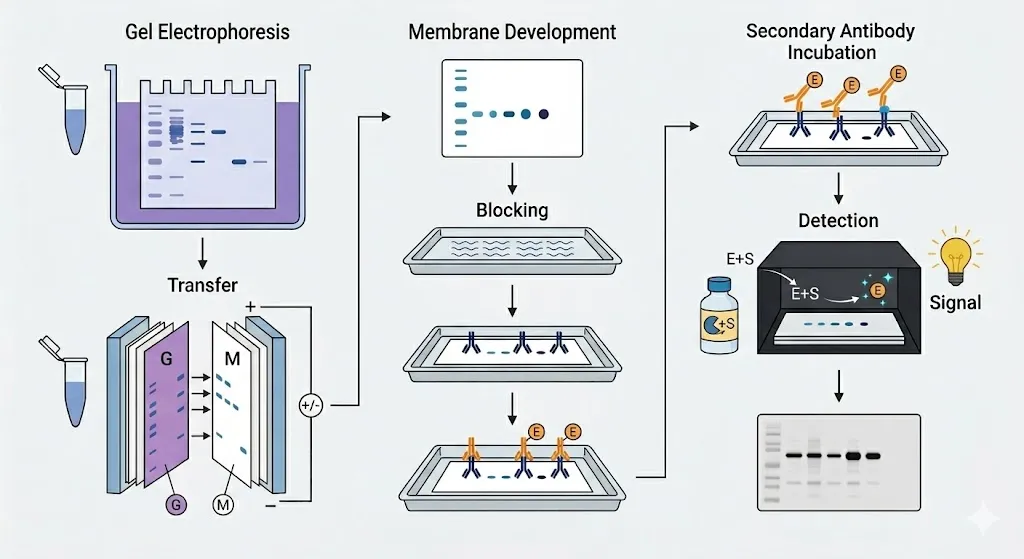
After the proteins have been separated by SDS-PAGE, they are still trapped inside the fragile gel. Western blotting moves them to a sturdy surface where they detected using antibodies.
- Transfer
The proteins are moved from the polyacrylamide gel onto a thin, durable membrane (usually nitrocellulose or PVDF). By applying an electric field perpendicular to the gel, the proteins are pushed out of the gel and stick firmly to the surface of the membrane, creating an exact replica of the original separation pattern.
- Blocking
The membrane is very sticky and will catch any protein it touches. To prevent antibodies from sticking to the empty spaces on the membrane (which would create a messy, dark background), the membrane is soaked in a “blocking” solution—often a dilute protein mix like non-fat dry milk or bovine serum albumin (BSA).
- Antibody Incubation
The membrane is treated with a Primary Antibody designed to recognize only the target protein. After washing away any unbound primary antibodies, a Secondary Antibody is added. This second antibody recognizes the first one and is usually attached to an enzyme or a fluorescent dye that acts as a signal.
- Detection
To see the results, a chemical substrate is added that reacts with the enzyme on the secondary antibody, producing light (chemiluminescence) or a colored deposit. This creates a dark band on a piece of film or a digital image, confirming not just the size of the protein, but its exact identity and how much of it is present.
