Acid-Base Titration is a fundamental quantitative laboratory technique used to determine the unknown concentration of a dissolved acid or base. By reacting a solution of unknown concentration with a solution of a precisely known concentration (the titrant), scientists can use the stoichiometry of the chemical reaction to calculate the exact molarity of the sample. This is the primary method used for everything from testing the acidity of wine to ensuring the potency of liquid antacids.
How Acid-Base Titration Works
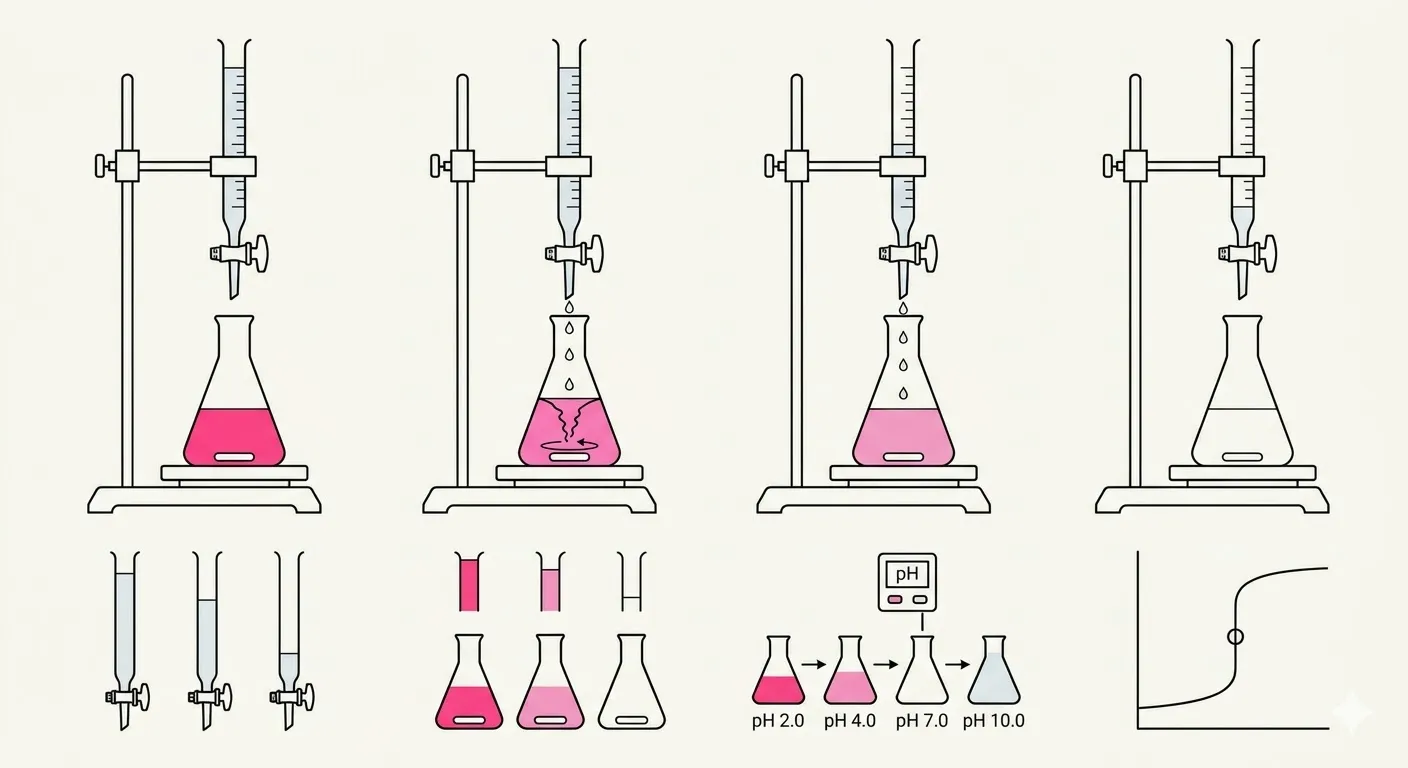
The process relies on a controlled chemical “battle” where the point of victory is reached when the acid and base have perfectly neutralized each other.
A precise volume of the unknown solution, called the analyte, is placed in an Erlenmeyer flask with a few drops of a pH indicator such as phenolphthalein, while a buret is filled with the titrant of known concentration. The stopcock of the buret is opened to allow the titrant to flow into the flask, where a neutralization reaction occurs — for example, titrating hydrochloric acid with sodium hydroxide produces water and salt: HCl + NaOH → H2O + NaCl. The goal is to reach the equivalence point, where the number of moles of added titrant is chemically equal to the number of moles of the substance in the unknown sample, meaning the acid and base have completely neutralized each other. Because the equivalence point is invisible, the end point is used instead — the moment the pH indicator permanently changes color, such as turning from clear to a very faint persistent pink, signaling the scientist to stop the flow immediately. By measuring exactly how many milliliters of titrant were used from the buret and knowing the titrant’s molarity, the concentration of the unknown can be calculated using the formula Macid × Vacid = Mbase × Vbase (assuming a 1:1 mole ratio; coefficients are added for other ratios).
resource: Lab Lexicon acid/base titration tool
