Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE) is a powerful laboratory technique used to separate proteins based exclusively on their molecular weight. Scientists often deal with “protein soups”—complex mixtures containing hundreds of different types of proteins. SDS-PAGE acts as a molecular sieve, allowing researchers to untangle this mixture to identify specific proteins, check the purity of a sample, or verify the size of a synthesized protein.
How SDS-PAGE Works
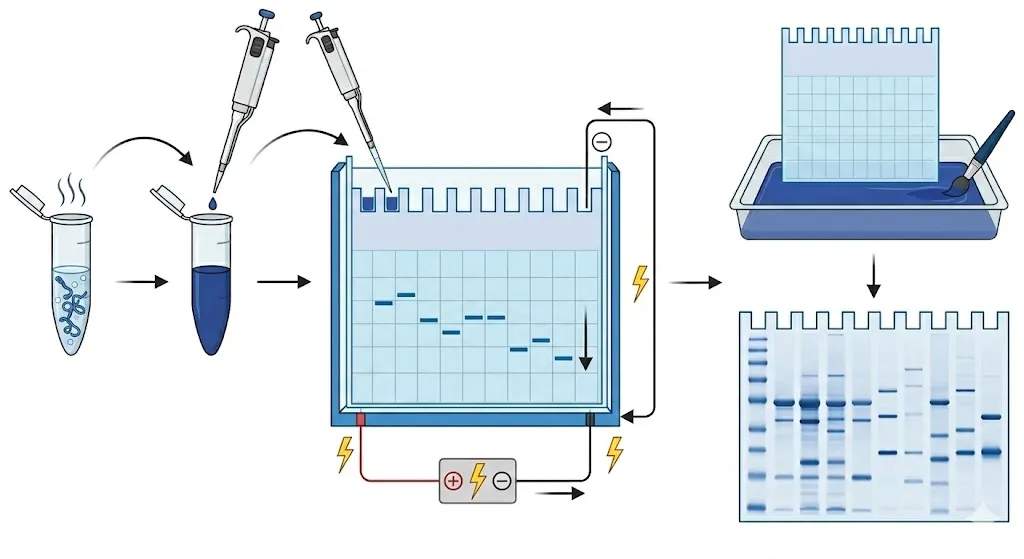
The process relies on the fact that proteins, unlike DNA, have complex, folded shapes and varying natural charges. To ensure size is the only factor influencing their movement, the experiment uses a series of steps to “reset” the protein properties.
- Denaturation
The protein sample is mixed with a buffer containing SDS (a strong detergent) and heated. The SDS acts as a chemical straightener; it unfolds the complex 3D structure of the proteins into linear chains. Crucially, the SDS coats the proteins in a uniform negative charge, ensuring they move toward the positive electrode regardless of their original charge.
- Loading
The sample is loaded into the wells of a polyacrylamide gel—a synthetic matrix that acts as a microscopic obstacle course. A “protein ladder” (a mixture of proteins with known molecular weights) is typically loaded into the first well to serve as a ruler for the rest of the experiment.
- Migration
An electric current is applied across the gel. Because the proteins are negatively charged, they move toward the anode (positive electrode) at the bottom. As they travel, smaller proteins navigate the pores of the gel with ease and move quickly, while larger proteins get tangled and move much more slowly.
- Visualization
After the elecctrophoresis is complete, the proteins are invisible to the naked eye. The gel is soaked in a stain—most commonly Coomassie Brilliant Blue. The dye binds to the proteins, revealing a series of distinct blue bands. By comparing the position of these bands to the protein ladder, scientists can calculate the exact molecular weight of each protein in the sample.
